Thank you to my friend and colleague, Dr. Benny Gavi, with whom we will be jointly sending out a weekly update. This will be based on our participation in Stanford “Town Halls” in both Internal Medicine and Emergency Medicine and other news we glean over the week. Our thoughts from this past week:
OVERALL
Coronavirus cases continue to expand in the Bay Area, however the sheltering in place is reducing the rate of expansion. The volume of hospitalized people in Northern California is gradually increasing (approximately doubling every week, see graphs below), however the volumes are currently manageable. It is expected that the number of people will increase and peak hospital utilization will occur in late April or early May. We may be sheltering in place until June. Overall, our local hospital, Stanford, appears to be well prepared.
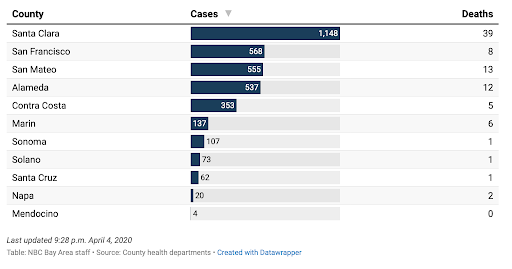
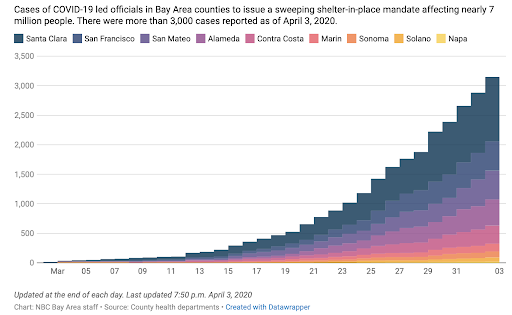
The Santa Clara Dashboard has been updated and also provides helpful data about current hospital and lab testing statistics.
WE’RE STILL STAYING HOME / #stayhomesavelives
As of March 31, Health officers in seven Bay Area jurisdictions are extending a previous stay-at-home order through May 3, 2020 in order to preserve critical hospital capacity across the region. The health officers have determined that more and stricter social distancing is needed to slow the rate of spread, prevent deaths, and stop the health care system from becoming overwhelmed. The new order adds some clarifying language around essential business and activities, as well as some new directives, including:
- Use of playgrounds, dog parks, public picnic areas, and similar recreational areas is prohibited. These areas must be closed to public use.
- Use of shared public recreational facilities such as golf courses, tennis and basketball courts, pools, and rock walls is prohibited. These facilities must be closed for recreational use.
- Sports requiring people to share a ball or other equipment must be limited to people in the same household
- Requires essential businesses to develop a social distancing protocol before April 3
- Most construction—residential and commercial—is prohibited
- Funerals limited to no more than 10 people attending
- Essential businesses expanded to include service providers that enable
residential transactions (notaries, title companies, Realtors, etc.); funeral homes and cemeteries; moving companies, rental car companies and rideshare services that specifically enable essential activities - Essential businesses that continue to operate facilities must scale down operations to their essential component only
PPE FOR HEALTH CARE PROVIDERS
A big boost for protective equipment was announced this week: utilizing UV light, one company in Massachusetts will be able to sterilize 80,000 NP 95 masks per day. More companies are expected to be able to do the same and will probably be available in California very soon.
TESTING
Home delivery of testing kits is temporarily delayed because of concerns by the FDA regarding quality of testing. Hopefully this can be resolved within the next week. Note there are two types of tests: one uses PCR (polymerase chain reaction) to look for actual coronavirus RNA to determine if you have COVID-19 at the time of testing while the other looks at antibodies in blood to determine if you HAD COVID-19 in the past.
Note also that there were some earlier questions about how accurate the testing was at Stanford (accuracy is measured by a test’s sensitivity and specificity). It appears that some of the early tests’ weakness had to do with how the samples were being collected rather than the test itself. More recently, samples being collected at Stanford have both outstanding sensitivity (96%) and specificity (nearly 100%), which means the test is quite good at determining whether or not you have COVID..
Lastly, here are some updates from the news this past week:
(3/27/20) Abbott launches a new test that can detect COVID-19 in as little as 5 minutes.
(3/26/20) Curative Inc labs racing to deliver 10,000 saliva tests / day to LA County.
(03/22/20) Everlywell offers COVID-19 test kit, but limiting it to frontline medical facilities.
SHOULD I WEAR A MASK IN PUBLIC? YES!
As discussed in prior updates, Coronavirus is contagious by droplets which land on surfaces. However, there is increasing evidence that Coronavirus is also spread by virus particles in the air. Therefore, there are increasing recommendations that people wear masks when in public. This is especially important if you are within 6 feet of another person for longer than a few seconds.
Masks reduce transmission by reducing droplets and aerosolized viral particles produced by an infected person (who may or may not know they are infected). Masks can also reduce the likelihood that an uninfected individual will inhale viral particles that are in the air produced by an infected person who was coughing, speaking or simply breathing.
In light of all of this, as of April 3rd, based on guidance from the California Department of Public Health, Bay Area health officials are now recommending residents cover their nose and mouth with cloth when leaving home for essential travel such as doctor appointments, grocery shopping or pharmacy visits. Examples of acceptable face coverings include bandanas, fabric masks, neck gaiters and scarves that can be easily washed with detergent and hot water. The covering needs to fit comfortably so that you are not tempted to adjust it. The public should NOT use medical masks as these should be preserved for first responders and health care workers. The hope is that these masks will remind people to keep physical distancing and limit the spread of disease. Still, the most important thing you can do to prevent the spread of COVID-19 is to stay home.
HYDROXYCHLOROQUINE FOR COVID TREATMENT?
A small trial published in China revealed that hydroxychloroquine may be helpful for mild or moderate disease. It was a small study of only 60 people and some American scientists question the validity
of the study. That said, much of the very early excitement has now been tampered down, but this hasn’t stopped the political pressure. Some claim that even our FDA put politics over science when it issued an emergency use authorization for chloroquine and hydroxychloroquine on March 29, 2020. On March 30 the University of Minnesota started a trial looking at hydroxychloroquine used as prophylaxis against COVID-19. So all to say, there is a lot to learn, a lot of political pressure and a tremendous amount of clinical need. The coming few weeks should be very interesting!
CONSIDER BUYING A FINGERTIP PULSE OXIMETER
A pulse oximeter is a device that is placed at the tip of the finger and easily measures oxygen level. Some people with COVID infection have found these devices helpful to track the severity of their disease or simply to assess whether they can stay home or if they should go to the hospital. We are learning that some people with COVID infection may have low oxygen levels (below 90-92%) because the virus can cause pneumonia, but people may not necessarily be short of breath. This device can be purchased on Amazon.
In health,
Eric Weiss & Benny Gavi


